Extra-Nodal Follicular Dendritic Cell Sarcoma with Unusual Morphological Features and PDL1 Expression
CC BY-NC-ND 4.0 ? Indian J Med Paediatr Oncol
DOI: DOI: 10.1055/s-0042-1742319
Abstract
Follicular dendritic cell (FDC) sarcoma, a rare tumor of FDCs, poses a diagnostic challenge due to its myriad morphologic and architectural patterns. We present a unique case of extra-nodal FDC sarcoma occurring in a 28-year-old gentleman who presented with multiple pelvic, peritoneal and retroperitoneal soft tissue, and axial skeletal lesions. The tumor was composed of islands and trabeculae of epithelioid cells with interspersed lymphocytes against a background of abundant myxoid matrix. On immunohistochemistry, in addition to the CD21, the tumor was positive for GATA3, inhibin, and programmed death-ligand 1 (PDL1), which have been rarely reported previously in FDC sarcoma. The patient underwent surgical excision of the tumor and was later offered immunotherapy. He has been disease-free for 2 years following immunotherapy. Our case highlights the potential role of immune checkpoint inhibitors in treating FDC sarcoma, which show immuno-expression of PDL1.
Keywords
follicular dendritic cell sarcoma - PDL1 - immunotherapyDeclaration of Patient Consent
The authors certify that they have obtained all appropriate patient consent forms.
Supplementary MaterialPublication History
26 April 2022 (online)
A-12, 2nd Floor, Sector 2, Noida-201301 UP, India
Abstract
Follicular dendritic cell (FDC) sarcoma, a rare tumor of FDCs, poses a diagnostic challenge due to its myriad morphologic and architectural patterns. We present a unique case of extra-nodal FDC sarcoma occurring in a 28-year-old gentleman who presented with multiple pelvic, peritoneal and retroperitoneal soft tissue, and axial skeletal lesions. The tumor was composed of islands and trabeculae of epithelioid cells with interspersed lymphocytes against a background of abundant myxoid matrix. On immunohistochemistry, in addition to the CD21, the tumor was positive for GATA3, inhibin, and programmed death-ligand 1 (PDL1), which have been rarely reported previously in FDC sarcoma. The patient underwent surgical excision of the tumor and was later offered immunotherapy. He has been disease-free for 2 years following immunotherapy. Our case highlights the potential role of immune checkpoint inhibitors in treating FDC sarcoma, which show immuno-expression of PDL1.
Keywords
follicular dendritic cell sarcoma - PDL1 - immunotherapy
Introduction
Follicular dendritic cell (FDC) sarcoma, first described by Monda et al,[1] is a tumor affecting both nodal and extra-nodal sites. It is presumed to originate from the benign FDCs.[2] FDCs are required for normal follicle formation and B-cell activation.[2] [3] FDC sarcoma mainly affects cervical, supraclavicular, axillary, mesenteric, and retroperitoneal lymph nodes.[4] [5] [6] Less than one-third cases occur in extra-nodal sites like oral cavity, tonsil, palate, nasopharynx, breast, pleura, gastrointestinal tract, liver, pancreas, spleen, soft tissue, pelvis, and peritoneum.[2] [4] [5] [6] FDC sarcoma can occur over a wide age range with a median age at diagnosis being 50 years without any gender predilection.[4] [6] [7] It is often a diagnostic conundrum due to morphologic overlap with many other tumors.[5] Of late, programmed death-ligand 1 (PDL1) immuno-expression has been noted in these tumors; however, the literature is sparse.[8] [9] [10] [11]
We, herein, present a morphologically and immunohistologically unusual case of extra-nodal FDC sarcoma in a young gentleman presenting as multiple intra-abdominal masses. We also discuss few reported PDL1 positive FDC sarcoma cases in literature while trying to assess the potential role of immunotherapy in treating these tumors.
Case Report
A 28-year-old gentleman presented with pain in his lower abdomen along with increased frequency of micturition for 2 to 3 days. An abdominal ultrasound performed as part of preliminary investigation revealed multiple intra-abdominal masses. On positron emission tomography and computed tomography (PET-CT) scan, a large, hypodense, low grade, fluorodeoxyglucose avid lesion was noted in the pelvic region measuring 9.2???9.4???12.8?cm (SUV max 4.57). Similar hypodense deposits were noted in the peritoneal and retroperitoneal soft tissues, in rectovesical pouch, and in axial skeleton. These findings were highly suspicious of a lymphoma.
We received four paraffin blocks of the peritoneal mass biopsy at our tertiary center for review. Microscopy showed a tumor composed of islands and trabeculae of cells separated by fibrous septae ([Fig. 1A], [B]) with few interspersed mature lymphocytes. Myxoid matrix was noted in the background of these cells ([Fig. 1C]). Individual cells were epithelioid to plasmacytoid with moderate amounts of eosinophilic cytoplasm, and enlarged nuclei with variable nucleoli ([Fig. 1D]). On immunohistochemistry (IHC), the tumor was focally positive for inhibin ([Fig. 2A]) and GATA3 ([Fig. 2B]) whereas negative for calretinin, WT1, and D240. Furthermore, AE1/AE3 and epithelial membrane antigen were also negative. MIB-1 labeling index was ?20%. Also,?p53?showed wild-type expression. Tumor cells were diffusely positive for CD21 ([Fig. 2C]) and CD23 ([Fig. 2D]). The tumor was mismatch repair protein proficient. Tumor cells showed strong and diffuse membranous staining for PDL1 ([Fig. 2E]). The patient underwent abdominal tumor excision along with resection of part of right distal colon, sigmoid colon, and rectum because of adhesions. Two months later, he was started on immunotherapy with PDL1 inhibitor, nivolumab. He responded well to immunotherapy and is disease-free at 2 years of follow-up
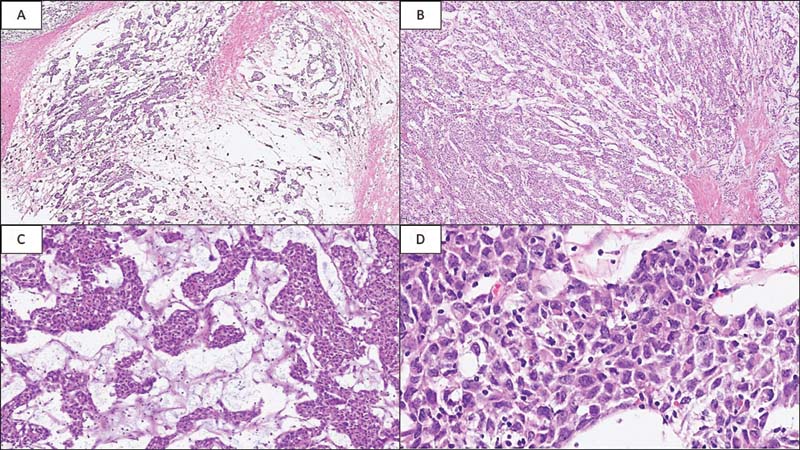
|?Fig. 1Photomicrograph of follicular dendritic cell sarcoma of peritoneum. (A) Loosely cohesive clusters of tumor cells separated by fibrous septae (hematoxylin and eosin, x20). (B) Islands of tumor cells in myxoid matrix (hematoxylin and eosin, x40). (C) Nests of plasmacytoid cells with eosinophilic cytoplasm intermixed with sparse lymphocytic infiltrate (hematoxylin and eosin, x100). (D) Polygonal cells with indistinct cell membranes, eosinophilic cytoplasm, moderate nuclear pleomorphism, vesicular chromatin, and variable nucleoli (hematoxylin and eosin, x400).
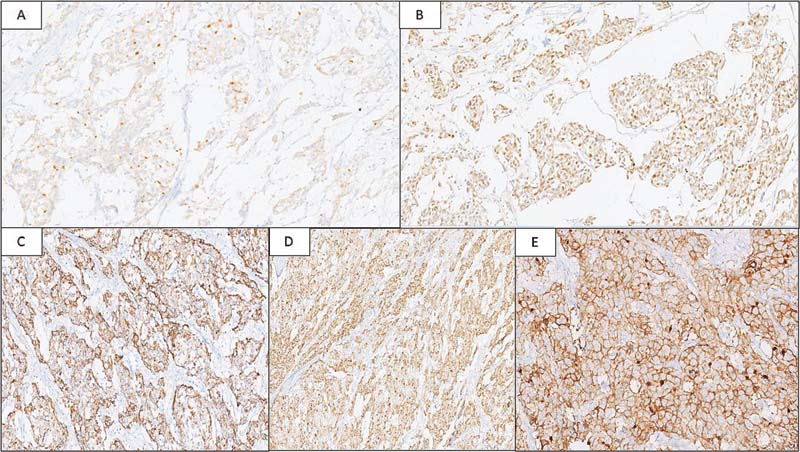
|?Fig. 2Tumor is focally positive for Inhibin (A: immunohistochemistry [IHC], x100) and GATA3 (B: IHC, x200), and shows diffuse positivity for CD21 (C: IHC, x200) and CD23 (D: IHC, x100). Strong and diffuse membranous positivity for programmed death-ligand 1 in tumor cells (E: IHC, x100).
Discussion
FDC sarcomas are rare tumors often misdiagnosed because of morphological overlap with other tumors, especially when presenting at extra-nodal sites. In addition to the syncytial islands of plasmacytoid/epithelioid cell pattern seen in our case, FDC sarcoma can present in a myriad of architectural patterns like storiform, whorled, fascicular, and diffuse sheets. Of these, the storiform pattern is the most common.[12] Individual cells show indistinct cell borders, moderate to abundant eosinophilic cytoplasm, and elongated to ovoid nuclei. In a subset of FDC sarcomas, high-grade histologic features are identified in the form of significant nuclear atypia, high mitotic count (11?35/10 high power fields), and extensive necrosis.[13] Histological variants of FDC sarcoma include folliculocentric B-cell-rich, inflammatory pseudotumor-like, and myxoid variants.[14] [15] [16] The folliculocentric B-cell-rich FDC sarcoma shows either macro-nodules composed predominantly of lymphocytes and tumor cells or small nodules of tumor cells surrounded by mantle of lymphocytes.[14] Inflammatory pseudotumor-like FDC sarcomas are associated with Epstein-Barr virus (EBV) infection and these are divided into two morphological variants, namely granulomatous and eosinophil rich.[15] Myxoid variant of FDC sarcoma shows prominent myxoid stroma and mucin pools.[16] As FDC sarcomas show morphologic overlap with many other conditions, IHC is an indispensable tool. Nodal and extra-nodal FDC sarcomas share immunophenotype and are usually positive for dendritic cell markers such as CD21, CD23, and/or CD35.[4] [5] Clusterin shows strong positivity in FDCs that helps to differentiate from other dendritic cell tumors in which it is weak or negative.[4] Podoplanin (D240) is another sensitive marker[4] and Gamma-Synuclein is a newer marker with high specificity.[17] Inhibin positivity has been reported in only two cases whereas GATA3, a transcription factor for the differentiation of breast epithelia, urothelium, and a subset of T cells, has been reported in only one case of FDC sarcoma in literature.[8] [18] Despite a sprinkling of lymphocytes in our case, an absence of plasma cells and eosinophils ruled out the possibility of inflammatory myofibroblastic tumor. Negativity for calretinin, WT1, and D240 ruled out a mesothelioma. Inhibin was focally positive; however, a negative calretinin stain ruled out the possibility of an adrenocortical carcinoma. In addition to the differential diagnoses discussed above, epithelioid extra-gastrointestinal stromal tumor (GIST) commonly seen in mesentery or omentum is another differential. On IHC, almost all the extra-intestinal GISTs show expression of c-kit, and around 50% show positivity for CD34.[19]
The clinical course of FDC sarcoma is variable with few cases showing aggressive clinical behavior. Nodal FDC sarcomas are often indolent, with low rate of metastases (?10%), as compared with extra-nodal ones, especially abdominal tumors, which have higher metastatic potential (>20%).[20] Though there is no uniform treatment strategy for FDC sarcoma, complete surgical resection is the primary treatment with or without adjuvant chemotherapy.[20] Intra-abdominal tumors require adjuvant chemotherapy because of their aggressive clinical behavior.[21]
With recent advancement in the field of immunotherapy, there has been a paradigm shift in oncology treatment practices. The success of immunotherapy lies on whether the tumor is immunologically ?hot or cold.? Generally, immune hot tumors are more amenable to immune checkpoint inhibitor (ICI) therapy.[22] Moreover, chemotherapy and radiotherapy have the potential of converting immunologically cold tumors to hot, thereby increasing the chances of success of immunotherapy.[23] This has been explored for tumors such as melanoma, non-small-cell lung cancer, colorectal cancer, and triple negative breast carcinoma.[22] [24] However, for FDC sarcoma, despite a clear immune cell infiltration, we do not perform PDL1 IHC as a standard practice. Also, PDL1-directed ICIs such as nivolumab and ipilimumab are not yet widely used for its primary treatment.[8] There are very few cases in literature where PDL1 has been performed ([Supplementary Table S1]).[8] [9] [10] [11] [25] In a study by Okada et al, 5 out of 10 cases were positive for PDL1.[9] Xu et al demonstrated PDL1 positivity in 10 out of 50 cases.[10] Of the 10 positive cases, 3 showed 20 to 59% tumor cell positivity whereas 7 showed >60% PDL1 positivity; intensity of staining varied from +2 to +3.[10] Agaimy et al demonstrated PDL1 positivity in 7 out of 15 cases.[11] However, immunotherapy was not offered as treatment to any of these patients.[9] [10] [11] Two cases reported by Lee et al expressing PDL1 showed benefit of immunotherapy.[8] Intensity of positivity in these two cases was +1 with scores of 60 to 70% and 10%, respectively. In both the cases, response to therapy was evaluated between 7 and 10 weeks by contrast enhanced CT or PET-CT, as well as by physical examination, at every treatment visit. The first patient showed recurrence after initial surgery and chemotherapy but had a complete radiological response after radiotherapy and ICI therapy, nivolumab, which was continued for 9 months, without disease recurrence. The second patient, a case of recurrent FDC sarcoma, had a partial response with ICI.[8] Cingam et al also described a case of FDC sarcoma wherein a trial of salvage nivolumab was attempted for liver metastases, but without any benefit.[26] In the present case, the patient was disease-free for 2 years after initiation of immunotherapy, which further clarifies the potential benefit of ICI in FDC sarcoma. This case highlights the difficulties in morphological diagnosis of FDC sarcoma and the value of ancillary IHC testing. Myxoid stroma is uncommon in FDC sarcoma, and many a times is not considered as a differential diagnosis. The expression of inhibin and GATA3 is extremely rare in FDC sarcoma and may cause diagnostic confusion to an unwary pathologist who might inadvertently label it as other diagnostic entity like adrenocortical malignancy (inhibin positive) or a urothelial malignancy (GATA 3 positive). The high PDL1 IHC expression and its value in clinical management with nivolumab add a lot of credibility to pathological predictive marker studies and biology of FDC sarcoma. Despite the strengths, our findings need to be confirmed by larger studies wherein they use ICI for the treatment of FDC sarcoma.
|
Sr no. |
Author and year |
Age range (years) |
Positive for PDL1/total cases |
PDL1 clone |
|---|---|---|---|---|
|
1 |
Okada et al (2020)[9] |
52?72 |
5/10 |
SP142, E1J2J, 28-8 |
|
2 |
Lee et al (2020)[8] |
40?60 |
2/2 |
SP263 |
|
3 |
Ren et al (2018)[25] |
67 |
0/1 |
Not documented |
|
4 |
Xu et al (2016)[10] |
Not documented |
10/50 |
405.9A11 |
|
5 |
Agaimy et al (2016)[11] |
21?75 |
7/15 |
28?8 |
|
6 |
Present case |
28 |
1/1 |
SP263 |
References
- onda L, Warnke R, Rosai J.?A primary lymph node malignancy with features suggestive of dendritic reticulum cell differentiation. A report of 4 cases. Am J Pathol 1986; 122 (03) 562-572
- rong>?Ioachim H, Medeiros L.?Granulocytic, histiocytic, and dendritic cell neoplasms. In: Ioachim's Lymph Node Pathology. 4th edition. Philadelphia, PA: Wolters Kluwer; 2009: 531-535
- rong>?van Nierop K, de Groot C.?Human follicular dendritic cells: function, origin and development. Semin Immunol 2002; 14 (04) 251-257
- rong>?Chan J, Pileri S, Fletcher C, Weiss L, Grogg K.?Follicular dendritic cell sarcoma. In: Swerdlow S, Campo E, Harris N, Jaffe ES, Pileri SA, Stein H, Thiele J. eds. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised. 4th edition. Lyon, France: IARC Press; 2017: 476-478
- rong>?Kaur R, Mehta J, Borges A.?Extranodal follicular dendritic Cell sarcoma-A review: ?what the mind does not know the eye does not see?. Adv Anat Pathol 2021; 28 (01) 21-29
- rong>?Li L, Shi YH, Guo ZJ. et al.?Clinicopathological features and prognosis assessment of extranodal follicular dendritic cell sarcoma. World J Gastroenterol 2010; 16 (20) 2504-2519
- rong>?Pang J, Mydlarz WK, Gooi Z. et al.?Follicular dendritic cell sarcoma of the head and neck: case report, literature review, and pooled analysis of 97 cases. Head Neck 2016; 38 (Suppl. 01) E2241-E2249
- rong>?Lee MY, Bernabe-Ramirez C, Ramirez DC, Maki RG.?Follicular dendritic cell sarcoma and its response to immune checkpoint inhibitors nivolumab and ipilimumab. BMJ Case Rep 2020; 13 (04) e234363
- rong>?Okada K, Takahara T, Suzuki Y. et al.?Histiocytic and dendritic cell neoplasms: reappraisal of a Japanese series based on t(14;18) and neoplastic PD-L1 expression. Pathol Int 2021; 71 (01) 24-32
- trong>?Xu J, Sun HH, Fletcher CD. et al.?Expression of programmed cell death 1 ligands (PD-L1 and PD-L2) in histiocytic and dendritic cell disorders. Am J Surg Pathol 2016; 40 (04) 443-453
- trong>?Agaimy A, Michal M, Hadravsky L, Michal M.?Follicular dendritic cell sarcoma: clinicopathologic study of 15 cases with emphasis on novel expression of MDM2, somatostatin receptor 2A, and PD-L1. Ann Diagn Pathol 2016; 23: 21-28
- trong>?Chan JKC.?Proliferative lesions of follicular dendritic cells. Adv Anat Pathol 1997; 4: 387-411
- trong>?Perez-Ordonez B, Erlandson RA, Rosai J.?Follicular dendritic cell tumor: report of 13 additional cases of a distinctive entity. Am J Surg Pathol 1996; 20 (08) 944-955
- trong>?Lorenzi L, Lonardi S, Petrilli G. et al.?Folliculocentric B-cell-rich follicular dendritic cells sarcoma: a hitherto unreported morphological variant mimicking lymphoproliferative disorders. Hum Pathol 2012; 43 (02) 209-215
- trong>?Li XQ, Cheuk W, Lam PW. et al.?Inflammatory pseudotumor-like follicular dendritic cell tumor of liver and spleen: granulomatous and eosinophil-rich variants mimicking inflammatory or infective lesions. Am J Surg Pathol 2014; 38 (05) 646-653
- trong>?Fisher C, Magnusson B, Hardarson S, Smith ME.?Myxoid variant of follicular dendritic cell sarcoma arising in the breast. Ann Diagn Pathol 1999; 3 (02) 92-98
- trong>?Zhang H, Maitta RW, Bhattacharyya PK. et al.??-Synuclein is a promising new marker for staining reactive follicular dendritic cells, follicular dendritic cell sarcoma, Kaposi sarcoma, and benign and malignant vascular tumors. Am J Surg Pathol 2011; 35 (12) 1857-1865
- trong>?Lavinia PM, Ryan WHL, Jeffrey M, Roberto NM.?Follicular dendritic cell sarcoma in the differential diagnosis of GATA3 positive tumors: a case report. Hum Pathol 2021; 23: 2214-3300
- trong>?Reith JD, Goldblum JR, Lyles RH, Weiss SW.?Extragastrointestinal (soft tissue) stromal tumors: an analysis of 48 cases with emphasis on histologic predictors of outcome. Mod Pathol 2000; 13 (05) 577-585
- trong>?Wu A, Pullarkat S.?Follicular dendritic cell sarcoma. Arch Pathol Lab Med 2016; 140 (02) 186-190
- trong>?Chan JK, Fletcher CD, Nayler SJ, Cooper K.?Follicular dendritic cell sarcoma. Clinicopathologic analysis of 17 cases suggesting a malignant potential higher than currently recognized. Cancer 1997; 79 (02) 294-313
- trong>?Maleki Vareki S.?High and low mutational burden tumors versus immunologically hot and cold tumors and response to immune checkpoint inhibitors. J Immunother Cancer 2018; 6 (01) 157
- trong>?Bonaventura P, Shekarian T, Alcazer V. et al.?Cold tumors: a therapeutic challenge for immunotherapy. Front Immunol 2019; 10: 168
- trong>?Emens LA.?Breast cancer immunotherapy: facts and hopes. Clin Cancer Res 2018; 24 (03) 511-520
- trong>?Ren W, Sun Q, Wu PY. et al.?Profiles of genomic alterations in primary esophageal follicular dendritic cell sarcoma: a case report. Medicine (Baltimore) 2018; 97 (48) e13413
- Cingam SR, Al Shaarani M, Takalkar A, Peddi P.?Follicular dendritic sarcoma masquerading as fibrosing mediastinitis. BMJ Case Rep 2017; bcr2016218889
Address for correspondence
Publication History
26 April 2022 (online)
A-12, 2nd Floor, Sector 2, Noida-201301 UP, India

|?Fig. 1Photomicrograph of follicular dendritic cell sarcoma of peritoneum. (A) Loosely cohesive clusters of tumor cells separated by fibrous septae (hematoxylin and eosin, x20). (B) Islands of tumor cells in myxoid matrix (hematoxylin and eosin, x40). (C) Nests of plasmacytoid cells with eosinophilic cytoplasm intermixed with sparse lymphocytic infiltrate (hematoxylin and eosin, x100). (D) Polygonal cells with indistinct cell membranes, eosinophilic cytoplasm, moderate nuclear pleomorphism, vesicular chromatin, and variable nucleoli (hematoxylin and eosin, x400).

|?Fig. 2Tumor is focally positive for Inhibin (A: immunohistochemistry [IHC], x100) and GATA3 (B: IHC, x200), and shows diffuse positivity for CD21 (C: IHC, x200) and CD23 (D: IHC, x100). Strong and diffuse membranous positivity for programmed death-ligand 1 in tumor cells (E: IHC, x100).
References
- ke R, Rosai J.?A primary lymph node malignancy with features suggestive of dendritic reticulum cell differentiation. A report of 4 cases. Am J Pathol 1986; 122 (03) 562-572
- oachim H, Medeiros L.?Granulocytic, histiocytic, and dendritic cell neoplasms. In: Ioachim's Lymph Node Pathology. 4th edition. Philadelphia, PA: Wolters Kluwer; 2009: 531-535
- an Nierop K, de Groot C.?Human follicular dendritic cells: function, origin and development. Semin Immunol 2002; 14 (04) 251-257
- han J, Pileri S, Fletcher C, Weiss L, Grogg K.?Follicular dendritic cell sarcoma. In: Swerdlow S, Campo E, Harris N, Jaffe ES, Pileri SA, Stein H, Thiele J. eds. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised. 4th edition. Lyon, France: IARC Press; 2017: 476-478
- aur R, Mehta J, Borges A.?Extranodal follicular dendritic Cell sarcoma-A review: ?what the mind does not know the eye does not see?. Adv Anat Pathol 2021; 28 (01) 21-29
- i L, Shi YH, Guo ZJ. et al.?Clinicopathological features and prognosis assessment of extranodal follicular dendritic cell sarcoma. World J Gastroenterol 2010; 16 (20) 2504-2519
- ang J, Mydlarz WK, Gooi Z. et al.?Follicular dendritic cell sarcoma of the head and neck: case report, literature review, and pooled analysis of 97 cases. Head Neck 2016; 38 (Suppl. 01) E2241-E2249
- ee MY, Bernabe-Ramirez C, Ramirez DC, Maki RG.?Follicular dendritic cell sarcoma and its response to immune checkpoint inhibitors nivolumab and ipilimumab. BMJ Case Rep 2020; 13 (04) e234363
- kada K, Takahara T, Suzuki Y. et al.?Histiocytic and dendritic cell neoplasms: reappraisal of a Japanese series based on t(14;18) and neoplastic PD-L1 expression. Pathol Int 2021; 71 (01) 24-32
- Xu J, Sun HH, Fletcher CD. et al.?Expression of programmed cell death 1 ligands (PD-L1 and PD-L2) in histiocytic and dendritic cell disorders. Am J Surg Pathol 2016; 40 (04) 443-453
- Agaimy A, Michal M, Hadravsky L, Michal M.?Follicular dendritic cell sarcoma: clinicopathologic study of 15 cases with emphasis on novel expression of MDM2, somatostatin receptor 2A, and PD-L1. Ann Diagn Pathol 2016; 23: 21-28
- Chan JKC.?Proliferative lesions of follicular dendritic cells. Adv Anat Pathol 1997; 4: 387-411
- Perez-Ordonez B, Erlandson RA, Rosai J.?Follicular dendritic cell tumor: report of 13 additional cases of a distinctive entity. Am J Surg Pathol 1996; 20 (08) 944-955
- Lorenzi L, Lonardi S, Petrilli G. et al.?Folliculocentric B-cell-rich follicular dendritic cells sarcoma: a hitherto unreported morphological variant mimicking lymphoproliferative disorders. Hum Pathol 2012; 43 (02) 209-215
- Li XQ, Cheuk W, Lam PW. et al.?Inflammatory pseudotumor-like follicular dendritic cell tumor of liver and spleen: granulomatous and eosinophil-rich variants mimicking inflammatory or infective lesions. Am J Surg Pathol 2014; 38 (05) 646-653
- Fisher C, Magnusson B, Hardarson S, Smith ME.?Myxoid variant of follicular dendritic cell sarcoma arising in the breast. Ann Diagn Pathol 1999; 3 (02) 92-98
- Zhang H, Maitta RW, Bhattacharyya PK. et al.??-Synuclein is a promising new marker for staining reactive follicular dendritic cells, follicular dendritic cell sarcoma, Kaposi sarcoma, and benign and malignant vascular tumors. Am J Surg Pathol 2011; 35 (12) 1857-1865
- Lavinia PM, Ryan WHL, Jeffrey M, Roberto NM.?Follicular dendritic cell sarcoma in the differential diagnosis of GATA3 positive tumors: a case report. Hum Pathol 2021; 23: 2214-3300
- Reith JD, Goldblum JR, Lyles RH, Weiss SW.?Extragastrointestinal (soft tissue) stromal tumors: an analysis of 48 cases with emphasis on histologic predictors of outcome. Mod Pathol 2000; 13 (05) 577-585
- Wu A, Pullarkat S.?Follicular dendritic cell sarcoma. Arch Pathol Lab Med 2016; 140 (02) 186-190
- Chan JK, Fletcher CD, Nayler SJ, Cooper K.?Follicular dendritic cell sarcoma. Clinicopathologic analysis of 17 cases suggesting a malignant potential higher than currently recognized. Cancer 1997; 79 (02) 294-313
- Maleki Vareki S.?High and low mutational burden tumors versus immunologically hot and cold tumors and response to immune checkpoint inhibitors. J Immunother Cancer 2018; 6 (01) 157
- Bonaventura P, Shekarian T, Alcazer V. et al.?Cold tumors: a therapeutic challenge for immunotherapy. Front Immunol 2019; 10: 168
- Emens LA.?Breast cancer immunotherapy: facts and hopes. Clin Cancer Res 2018; 24 (03) 511-520
- Ren W, Sun Q, Wu PY. et al.?Profiles of genomic alterations in primary esophageal follicular dendritic cell sarcoma: a case report. Medicine (Baltimore) 2018; 97 (48) e13413
- Cingam SR, Al Shaarani M, Takalkar A, Peddi P.?Follicular dendritic sarcoma masquerading as fibrosing mediastinitis. BMJ Case Rep 2017; bcr2016218889


 PDF
PDF  Views
Views  Share
Share

