Round Cell Tumors: Classification and Immunohistochemistry
CC BY-NC-ND 4.0 · Indian J Med Paediatr Oncol 2017; 38(03): 349-353
DOI: DOI: 10.4103/ijmpo.ijmpo_84_16
Abstract
Round cell tumors as the name suggest are comprised round cells with increased nuclear-cytoplasmic ratio. This group of tumor includes entities such as peripheral neuroectodermal tumor, rhabdomyosarcoma, synovial sarcoma, non-Hodgkin's lymphoma, neuroblastoma, hepatoblastoma, Wilms' tumor, and desmoplastic small round cell tumor. These round cells tumors are characterized by typical histological pattern, immunohistochemical, and electron microscopic features that can help in differential diagnosis. The present article describes the classification and explains the histopathology and immunohistochemistry of some important round cell tumors.
Publication History
Article published online:
04 July 2021
© 2017. Indian Society of Medical and Paediatric Oncology. This is an open access article published by Thieme under the terms of the Creative Commons Attribution-NonDerivative-NonCommercial-License, permitting copying and reproduction so long as the original work is given appropriate credit. Contents may not be used for commercial purposes, or adapted, remixed, transformed or built upon. (https://creativecommons.org/licenses/by-nc-nd/4.0/.)
Thieme Medical and Scientific Publishers Pvt. Ltd.
A-12, 2nd Floor, Sector 2, Noida-201301 UP, India
Abstract
Round cell tumors as the name suggest are comprised round cells with increased nuclear-cytoplasmic ratio. This group of tumor includes entities such as peripheral neuroectodermal tumor, rhabdomyosarcoma, synovial sarcoma, non-Hodgkin's lymphoma, neuroblastoma, hepatoblastoma, Wilms' tumor, and desmoplastic small round cell tumor. These round cells tumors are characterized by typical histological pattern, immunohistochemical, and electron microscopic features that can help in differential diagnosis. The present article describes the classification and explains the histopathology and immunohistochemistry of some important round cell tumors.
Introduction
The term round cell tumor describes a group of highly aggressive malignant tumors composed of relatively small and monotonous undifferentiated cells with increased nuclear-cytoplasmic ratio.[1] Malignant small round cell tumors (MSRCT) is a term used for tumors composed of malignant round cells that are slightly larger or double the size of red blood cells in air-dried smears.[2]
This group of neoplasms is characterized by small, round, relatively undifferentiated cells. Differential diagnosis of small round cell tumors is particularly difficult due to their undifferentiated or primitive character. Tumors that show good differentiation are generally easy to diagnose, but identification of the diagnostic, morphological features is difficult when a tumor is poorly differentiated, therefore, no definitive diagnosis may be possible.[3] Fine-needle aspiration cytology (FNAC) plays an important role in the diagnosis of these tumors.[2,4,5]
Classification
On the basis of round cell pattern
- Diffuse round cell pattern
- Ewing's sarcoma
- Primitive neuroectodermal tumor (PNET)
- Merkel cell carcinoma
- Embryonal rhabdomyosarcoma (ERMS)
- Small cell carcinoma
- Lymphoma
- Leukemic infiltrate.
- Septate or lobulated round cell pattern
- Small round cells are divided by fibrous/fibrovascular septate
- Ewing's sarcoma
- Alveolar rhabdomyosarcoma (ARMS).
- Alveolar/pseudoalveolar round cell pattern
This pattern includes focal, poor cohesion of the round cell population resulting in pseudo alveolar appearance
- ARMS
- PNET.
- Round cell pattern with rosettes
A rosette' is like a flower, with the cells being arranged radially around a central area
- Flexner's (also called Flexner – Winterstein, true rosettes) – contain clearly delineated empty central lumen
e.g., neuroblastoma, PNET
- Homer Wright rosette-center has no lumen, but abundant fibrillary material
e.g., neuroblastoma.
- Round cell pattern with hemangiopericytomatous vascular pattern
e.g., poorly differentiated synovial sarcoma, Mesenchymal chondrosarcoma.
- Round cell pattern with other components
- Pseudo glands – poorly differentiated synovial sarcoma
- Cartilage – mesenchymal chondrosarcoma.
According to size of round cell
- Small round cell – Squamous cell carcinoma, PNET, Ewing's sarcoma, melanoma, rhabdomyosarcoma (RMS), Langerhans cell disease, lymphoma, adenocarcinoma, neuroendocrine carcinoma, Merkel cell carcinoma, olfactory neuroblastoma
- Large round cell – Squamous cell carcinoma, adenocarcinoma, melanoma, RMS, lymphoid tumors, paraganglioma.
On the basis of origin
- Neurogenic origin: Ewing's sarcoma/PNET, neuroblastoma, retinoblastoma, medulloblastoma, Merkel cell tumor, paragangliomas, small cell tumor of lung
- Mesenchymal origin
- Myogenic differentiation
- ERMS
- ARMS.
- Osteoid differentiation
- Small cell osteosarcoma.
- Chondroid differentiation
- Mesenchymal chondrosarcoma.
- Adipose tissue like differentiation
- Myxoid/round cell liposarcoma.
Hematolymphoid origin
- Lymphoma/”reticulum cell sarcoma.”
Malignant soft tissue tumors of uncertain type
- Desmoplastic small round cell tumor (DSRCT)
- Poorly differentiated synovial sarcoma.
Ewing's Sarcoma and Primitive Neuroectodermal Tumor
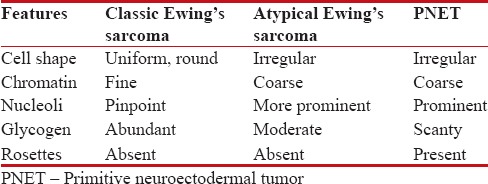
Ewing's sarcoma is a sarcoma of bone classically described under small round cell tumors. There is considerable clinical and histologic overlap between this tumor and the PNET. Ewing's sarcoma arises within the bone, but can also occur within the soft tissue (extraosseous Ewing's sarcoma) and PNET arises within soft tissues.[6] This neoplasm mainly affects the pelvis and the femur region and predominates in the second decade of life [Table 1].
Table 1
Histological characteristics of Ewing Sarcoma and PNET

|
PNET is a small round cell malignancy of primitive, neuroectodermal tissue or pluripotential, migratory neural crest cells that arises from the soft tissue or bone, commonly affecting older children and adults.[7]
The term, “PNET” includes MSRCTs of the thoracopulmonary region (Askin's tumor), extraskeletal Ewing's sarcoma, peripheral neuroblastoma, and peripheral neuroepithelioma.[2,8]
FNAC reveals the presence of tumor cells that are arranged in relatively small, tight clusters with the presence of round or irregular nuclei lacking nucleoli. These small blue cells have a high nucleocytoplasmic ratio. Two population of cells have been described large chief cells and smaller dark cells.[2]
The cytoplasm of these cells is pale blue and contains variable numbers of punched-out vacuoles which correspond to glycogen deposits, can be well demonstrated by periodic acid–Schiff (PAS) staining. However, the presence of large amounts of intracellular glycogen is not a specific finding as while up to 35% of all Ewing's sarcoma cases do not contain detectable glycogen, many other childhood tumors do contain detectable glycogen.[2]
The tumor shows variable numbers of pseudorosettes; fibrillary matrix and Homer Wright rosettes are seen at times, and mitotic figures are rarely detected.
Radiographic features
Onionskin or sunburst appearance.
Special stains
PAS with diastase (glycogen present in 75% of cases), immunohistochemistry.
Neuroblastoma
It is the third most common malignant extracranial solid tumor of childhood. Neuroblastoma and its related variants are derived from primitive neural crest cells that migrate from the mantle layer of the developing spinal cord and populate the primordial of the sympathetic ganglia and adrenal medulla [Table 2].[9]
Table 2
Histological characteristics of Neuroblastoma and variants
|
Rhabdomyosarcoma
RMS is the most commonly found soft tissue sarcoma in children wherein the cancer cells are thought to arise from skeletal muscle progenitors. These tumors are currently classified into ERMS, ARMS, and pleomorphic RMS (PRMS) subtypes.[9]
Subtypes
- ERMS: Mostly affects the children younger than 10 years of age. Features include:
- Varying degree of cellularity with alternating densely packed hypercellular areas and loosely textured myxoid areas
- A matrix containing little collagen and varying amount of myxoid material.
- ARMS: Composed of large aggregates of poorly differentiated round or oval tumor cells that show central loss of cellular cohesion and formation of irregular alveolar spaces. The individual cellular aggregates are separated and surrounded by dense hyalinized fibrous septa
Cells at the periphery of alveolar spaces adhere in a single layer to the fibrous septa while the cells at the center of the alveolar spaces are loosely arranged or free floating.[11]
- PRMS: Composed of spindle-shaped cells arranged in a fascicular pattern with eosinophilic stringy cytoplasm.
Special stains
- PAS with or without diastase: For intracellular glycogen
- Colloidal iron and alcian blue: For extracellular mucinous material
- Masson's trichrome, phosphotungstic acid hematoxylin.
Desmoplastic Small Round Cell Tumor[12]
DSRCT is a rare neoplasm that was first described by Gerald and Rosai in 1989.[12,13] It is a high-grade tumor that mostly affects abdominal cavity and visceral organs. It differs from other childhood tumors due to its clinical features, morphology, and its immunohistochemistry staining pattern.[2,14] Tumors are composed of sharply demarcated nests of varying size with small round or oval cells embedded in the hypervascular desmoplastic stroma. Large tumor cell nests have central necrosis [Table 3].[15]
Table 3
Immunohistochemical Data on Desmoplastic Round Cell Tumor

| The arrangement of the cells
- Large nests with central necrosis
- Tubular like structures
- Trabeculae separated by fibrovascular septa reminiscent of a “Zellballen” pattern.
Other features include
- Tumor cells have cleared out cytoplasm or a signet ring appearance
- Rhabdoid like foci in which tumor cells have paranuclear intracytoplasmic hyaline inclusions composed of aggregates of intermediate filaments
- Wright like rosettes.
Immunohistochemical Data On Desmoplastic Round Cell Tumors[15]

|
Willm's Tumor
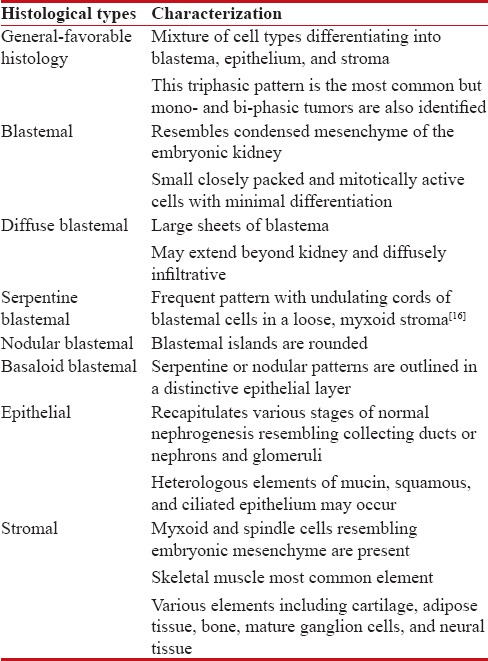
Willm's tumor (WT) or nephroblastoma is the cancer of kidneys that typically occurs in children rarely in adults [Table 4].
Table 4
Histological variants of Willm's tumor
|
Markers for Wilm's tumor
Cytokeratin, Desmin, WT-1 protein expression, NB84, CCN-3 protein.[17]
Immunohistochemistry of Round Cell Tumors
Markers:[9]
- CD 99
- CD 99 is a transmembrane glycoprotein of 30–32 KDa
- It plays a role in cellular adhesion and regulation of cellular proliferation
- Normal tissue that commonly displays strong expression of CD99 include:
- Cortical thymocytes
- Sertoli cells
- Endothelium
- Pancreatic islets
- Ependyma
- Epithelium (urothelium, squamous epithelium, columnar epithelium)
- It is specific for:
- Ewing's sarcoma – 90%
- Lymphoblastic lymphoma – 90%
- Synovial sarcoma – >75%
- Mesenchymal chondrosarcoma – 50%
- Osteosarcoma and desmoplastic round cell tumor – Rare
- Neuroblastoma – Never reported (-).
- NB 84
- It is sensitive marker for neuroblastoma (75%), Ewing's sarcoma (16%–25%)
- Also positive for RMS, WT, osteosarcoma, desmoplastic round cell tumor.
- S-100
- It is a marker for benign and malignant nerve sheath tumors
- Composed of two subunits α and β that combine to form 3 isotypes: α-α isotype found in the myocardium, skeletal muscle and neurons, α-β isotype found in chondrocytes, glia and skin adnexae, β-β isotype found in Langerhans and Schwann cells.
- Desmin
- It is the intermediate filament protein associated with both smooth and skeletal muscle differentiation
- Rarely expressed by myofibroblasts and their corresponding tumors
- In skeletal muscles desmin is localized to Z-zone between myofibrils when it serves as binding material for contractile apparatus. In smooth muscles, it is associated with cytoplasmic dense bodies
- Desmin can also be expressed by nonmuscle cells including fibroblastic reticulum cells of lymph nodes, submesothelial fibroblast, and endometrial stromal cells
- Expressed in PNET, desmoplastic round cell tumors, neuroblastoma, mesothelial cells and tumors, WT.
- Cytokeratins
- Used for distinguishing epithelial from nonepithelial tumors (lymphomas, sarcomas, melanomas)
- Expressed in: Carcinoma, epithelial sarcoma, leiomyosarcoma, mesothelioma
- Also expressed by round cell tumors such as Ewing's sarcoma/PNET, RMS, WT, desmoplastic round cell tumor.
- Myogenic transcription factor
- Help in the differentiation of mesenchymal progenitor cells to myogenic lineage and subsequent maintenance of the skeletal muscle phenotype.
- PAX 5
- PAX 5 is a member of the paired box transcription factors involved in the development and is expressed in hematopoietic malignancies of B-cell lineage
- Expressed in cases of neuroendocrine carcinomas, urothelial tumors, Merkel cell carcinoma, glioblastoma, and neuroblastoma
Table 5
Screening for undifferentiated round cell tumors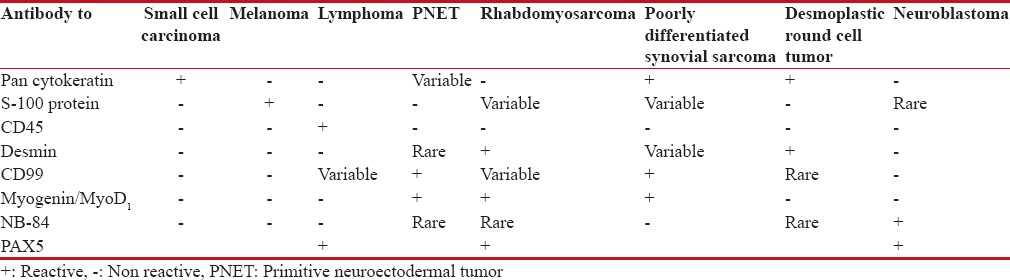
|
Conclusion
The ubiquitous distribution and diverse histology of different round cell tumors pose a challenge in their diagnosis. The diagnostic aids like the use of special stain, immunocytochemistry, flow cytometric immunophenotyping, and reverse-transcriptase polymerase chain reaction help to differentiate and diagnose these group of tumors. The early diagnosis of these tumors implicates the appropriate therapeutic modalities, including neo-adjuvant chemotherapy in advanced malignancy.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Devoe K, Weidner N. Immunohistochemistry of small round-cell tumors. Semin Diagn Pathol 2000;17:216-24.
- Rajwanshi A, Srinivas R, Upasana G. Malignant small round cell tumors. J Cytol 2009;26:1-10.
- Akhtar M, Ali MA, Sabbah R, Bakry M, Nash JE. Fine-needle aspiration biopsy diagnosis of round cell malignant tumors of childhood. A combined light and electron microscopic approach. Cancer 1985;55:1805-17.
- McGahey BE, Moriarty AT, Nelson WA, Hull MT. Fine-needle aspiration biopsy of small round blue cell tumors of childhood. Cancer 1992;69:1067-73.
- Layfield LJ, Liu K, Dodge RK. Logistic regression analysis of small round cell neoplasms: A cytologic study. Diagn Cytopathol 1999;20:271-7.
- Rajendran A, Sivapathasundharam B. Shafer's Textbook of Oral Pathology. 7th ed. New Delhi: Elsevier; 2012.
- Das D, Kuri GC, Deka P, Bhattacharjee K, Bhattacharjee H, Deka AC. Primary primitive neuroectodermal tumor of the orbit. Indian J Ophthalmol 2009;57:391-3.
- Silverman JF, Berns LA, Holbrook CT, Neill JS, Joshi VV. Fine needle aspiration cytology of primitive neuroectodermal tumors. A report of these cases. Acta Cytol 1992;36:541-50.
- Goldblum JR, Weiss SW. Enzinger and Weiss's Soft Tissue Tumors. 4th ed. Canada: Mosby; 2001.
- Wenig BM. Atlas of Head and Neck Pathology. 3rd ed. Philadelphia: Elsevier; 2015.
- Odoi AT, Dassah ET, Darkey DE, Owusu-Afriyie O, Valkov AY. Advanced alveolar rhabdomyosarcoma of the uterus: A case report. Afr J Reprod Health 2009;13:167-73.
- Chang F. Desmoplastic small round cell tumors: Cytologic, histologic, and immunohistochemical features. Arch Pathol Lab Med 2006;130:728-32.
- Hayes-Jordan A, Anderson PM. The diagnosis and management of desmoplastic small round cell tumor: A review. Curr Opin Oncol 2011;23:385-9.
- Akhtar M, Iqbal MA, Mourad W, Ali MA. Fine-needle aspiration biopsy diagnosis of small round cell tumors of childhood: A comprehensive approach. Diagn Cytopathol 1999;21:81-91.
- Barnoud R, Sabourin JC, Pasquier D, Ranchère D, Bailly C, Terrier-Lacombe MJ, et al. Immunohistochemical expression of WT1 by desmoplastic small round cell tumor: A comparative study with other small round cell tumors. Am J Surg Pathol 2000;24:830-6.
- Mostofi FK, Davis CJ Jr. Histological Typing of Kidney Tumours: In Collaboration with L. H. Sobin and Pathologist in 6 countries. Springer Berlin Heidelberg; 1998.
- Subramaniam MM, Lazar N, Navarro S, Perbal B, Llombart-Bosch A. Expression of CCN3 protein in human Wilms' tumors: Immunohistochemical detection of CCN3 variants using domain-specific antibodies. Virchows Arch 2008;452:33-9.
- Sullivan LM, Atkins KA, LeGallo RD. PAX immunoreactivity identifies alveolar rhabdomyosarcoma. Am J Surg Pathol 2009;33:775-80.
References
- Devoe K, Weidner N. Immunohistochemistry of small round-cell tumors. Semin Diagn Pathol 2000;17:216-24.
- Rajwanshi A, Srinivas R, Upasana G. Malignant small round cell tumors. J Cytol 2009;26:1-10.
- Akhtar M, Ali MA, Sabbah R, Bakry M, Nash JE. Fine-needle aspiration biopsy diagnosis of round cell malignant tumors of childhood. A combined light and electron microscopic approach. Cancer 1985;55:1805-17.
- McGahey BE, Moriarty AT, Nelson WA, Hull MT. Fine-needle aspiration biopsy of small round blue cell tumors of childhood. Cancer 1992;69:1067-73.
- Layfield LJ, Liu K, Dodge RK. Logistic regression analysis of small round cell neoplasms: A cytologic study. Diagn Cytopathol 1999;20:271-7.
- Rajendran A, Sivapathasundharam B. Shafer's Textbook of Oral Pathology. 7th ed. New Delhi: Elsevier; 2012.
- Das D, Kuri GC, Deka P, Bhattacharjee K, Bhattacharjee H, Deka AC. Primary primitive neuroectodermal tumor of the orbit. Indian J Ophthalmol 2009;57:391-3.
- Silverman JF, Berns LA, Holbrook CT, Neill JS, Joshi VV. Fine needle aspiration cytology of primitive neuroectodermal tumors. A report of these cases. Acta Cytol 1992;36:541-50.
- Goldblum JR, Weiss SW. Enzinger and Weiss's Soft Tissue Tumors. 4th ed. Canada: Mosby; 2001.
- Wenig BM. Atlas of Head and Neck Pathology. 3rd ed. Philadelphia: Elsevier; 2015.
- Odoi AT, Dassah ET, Darkey DE, Owusu-Afriyie O, Valkov AY. Advanced alveolar rhabdomyosarcoma of the uterus: A case report. Afr J Reprod Health 2009;13:167-73.
- Chang F. Desmoplastic small round cell tumors: Cytologic, histologic, and immunohistochemical features. Arch Pathol Lab Med 2006;130:728-32.
- Hayes-Jordan A, Anderson PM. The diagnosis and management of desmoplastic small round cell tumor: A review. Curr Opin Oncol 2011;23:385-9.
- Akhtar M, Iqbal MA, Mourad W, Ali MA. Fine-needle aspiration biopsy diagnosis of small round cell tumors of childhood: A comprehensive approach. Diagn Cytopathol 1999;21:81-91.
- Barnoud R, Sabourin JC, Pasquier D, Ranchère D, Bailly C, Terrier-Lacombe MJ, et al. Immunohistochemical expression of WT1 by desmoplastic small round cell tumor: A comparative study with other small round cell tumors. Am J Surg Pathol 2000;24:830-6.
- Mostofi FK, Davis CJ Jr. Histological Typing of Kidney Tumours: In Collaboration with L. H. Sobin and Pathologist in 6 countries. Springer Berlin Heidelberg; 1998.
- Subramaniam MM, Lazar N, Navarro S, Perbal B, Llombart-Bosch A. Expression of CCN3 protein in human Wilms' tumors: Immunohistochemical detection of CCN3 variants using domain-specific antibodies. Virchows Arch 2008;452:33-9.
- Sullivan LM, Atkins KA, LeGallo RD. PAX immunoreactivity identifies alveolar rhabdomyosarcoma. Am J Surg Pathol 2009;33:775-80.


 PDF
PDF  Views
Views  Share
Share

